N.S. Sundaresan
The teaching of science to young students particularly at the middle school level can be quite a challenge. Especially since this is where they are introduced to science as a subject. New facts and some theory about natural phenomena have to be introduced at this stage at least in a general way.
In this scenario the teacher is quite aware that some experiments have to be shown in order to keep the interest of the children alive. However, not every concept can be explained with an experiment due to lack of time. Hence the need to be selective in choosing the topics for experiments.
With this in mind I have collected a few experiments, described below, which pertain to the concepts relating to ‘matter’. The experiments are chosen so that even a teacher in a not so well-equipped school will be able to perform them with materials available at hand. These are well-known experiments, which are often given in textbooks. However, it is hoped that the contextual difference in the way these are suggested will be useful.
Concepts about matter have to be introduced in a general but convincing way. Some elementary idea has to be given about particles constituting matter. It is suggested that the following points could be included in your discussion with the students. The smallest particles constituting matter are atoms, which contain positive and negative charges within them. It will be helpful to summarize the forces operating inside matter thus – the atoms are held together by strong forces called chemical bonds which are electrical in nature. The aggregate of atoms, namely molecules, are held together by weak forces called intermolecular forces. All forces operating inside matter are composed of only these strong and weak forces which are electrical in nature. These forces vary from substance to substance and hence the strength with which these particles are bound also varies from substance to substance. Thus we have hard crystals, soft powders, liquids (low and high boiling) and gases. Also, the change from one physical state to another depends on the strength of the intermolecular forces.
Energy is released when matter is formed, but energy must be supplied to break matter (e.g., crystals to ions, solids to liquids, etc.). Chemical bonds can be broken by supplying a large amount of energy (heat, for example). Solubility of substances also depends upon overcoming one set of intermolecular forces by another set. All these have to be kept as a central idea while describing natural phenomena.
Prior to showing experiments it would be useful to show what type of matter we are interested in. This can be illustrated with chemical and biological samples with some substances which should include solids, liquids and gases. Generally, only chemical samples such as metallic or non-metallic solids are used in experiments, however, biological materials like vegetables and fruits (potato, orange, banana) and gases with smell could also be tried. These type of samples will serve to show the generality of matter. This is to complement or enlarge the students’ earlier learning about elements, mixtures and compounds.
A. Solubility of matter
An interesting property of matter is the ability of some substances to dissolve some others. The solubility of substances can be used as a phenomenon to kindle the interest of the students. Many interesting effects can be observed while dissolving a substance in a solvent. However, the explanation of the theory behind the observations is quite complex. Therefore, the teacher can, at this stage, give only a limited explanation mentioning intermolecular forces and polarity of substances.
For example, water will dissolve ionic substances such as salts since water is polar in nature. However, some organic substances (not all) may not dissolve in water but dissolve in substances like benzene. The substance present in smaller quantity is called solute and the one in larger quantity is called solvent. Dissolving leads to a homogeneous mixture which is called a solution. Formation of a solution is sometimes accompanied by either release of heat or absorption of heat. This leads to heating or cooling of the solution. All these involve overcoming many attractive forces and hence cause energy changes.
Experiment 1: To show how one substance dissolves another
Objective: This experiment is intended to show what happens when a solid matter is added to a liquid.
Materials
• Distilled water
• Common salt
• Sugar
• Calcium chloride
• Naphthalene (powder)
• Alcohol (rectified spirit), benzene
• A sensitive thermometer (0 to 110° C)
Procedure and observations
i. Take about 50 ml of water in a beaker. Add small quantities of common salt carefully stirring until no more can dissolve. If possible, use a sensitive thermometer (0.1° C graduation) in the beaker. While stirring note changes, if any, in the temperature.
ii. Repeat the experiment with calcium chloride instead of common salt.
iii. Repeat with alcohol and common salt in place of water and common salt.
iv. Repeat the experiment with water and naphthalene powder.
v. Repeat the experiment with benzene and naphthalene powder.
Questions for students
- Why does common salt dissolve in water only?
- Was there any temperature change during the process of dissolving salt in water?
- Why does naphthalene powder dissolve only in benzene and not in water?
- What was the temperature change when calcium chloride was dissolved in water?
Water is a polar molecule which means charges are present on the surface of the molecule. Other solvents like benzene are nonpolar. As a general rule, nonpolar substances dissolve only in nonpolar solvents just as ionic substances dissolve only in polar solvents.
Sodium chloride is an ionic solid. Hence there is an attraction for the sodium ion in the common salt leading to the breaking of the sodium and chlorine bond. The water then surrounds the sodium ion and to some extent the chloride ion.
In the case of sodium chloride there is absorption of energy from the water and hence the solution feels cold. In the case of calcium chloride, there is a net energy release and hence the solution becomes warm.
B. Change of state in matter
The physical state of matter such as being a solid, liquid or gas is dependent upon the binding forces between the molecules or atoms of the substance. Since particles come closer to each other by losing energy, conversely energy has to be supplied to keep particles away from each other. Thus, heat energy has to be supplied to melt a solid, to boil it to form a vapour or gas, or to sublime a solid. Since these are physical changes they can be reversed. By removing energy, you can condense a vapour or freeze a liquid.
The two experiments given below can be used to illustrate the ideas mentioned above.
Experiment 2: Distillation of a mixture of solid in liquid.
Objective: To show that by supplying energy selectively we can separate a solid-liquid mixture.
Materials
• Common salt
• Water
• Alcohol (or surgical spirit available in medical stores)
• Small clear glass bottle with cap
• A drinking straw
• A glass beaker large enough to keep the glass bottle
• Water soluble ink or water colour pigment
• Tripod stand and gauze
Procedure and observations
Take about 20 ml of alcohol in the glass bottle. Add a few drops of ink. Make a hole in the cap large enough to insert the drinking straw. Insert the straw to about half the distance from the top of the bottle. Wrap the straw with a piece of wet cloth. Close the bottle with the cap. (See Figure 1)
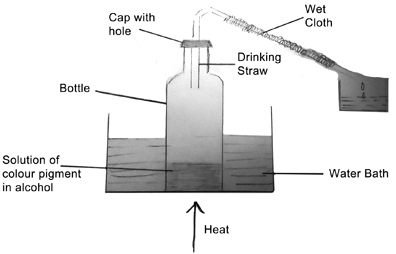
Bend the straw at an inch from the top to give an angle of about 45°. Take about 50 ml of water in the glass beaker. Place the bottle in the beaker, keep the beaker on a tripod stand with a wire gauze under the beaker. Heat from below. The heating should continue even if the water in the beaker boils. After sometime you should see drops of alcohol from the bottle dripping down from the straw. Keep a small container to collect the drops. Heat till all the alcohol in the bottle has boiled off. Observe if any residue is left in the bottle.
Questions for students
- Why do alcohol and water boil at different temperatures?
- Why did the ink not come along with the alcohol?
Note: The teacher should mention how in this experiment the liquid molecules are less firmly held together and hence boil off leaving the solid behind. This property is used for separation. You may have to add a larger volume of water in the beaker. The idea is to immerse the bottle to about half its height.
The experiment could be tried with a small quantity of common salt in water. The manufacture of salt from sea water in salt pans could be mentioned here.
Experiment 3: Sublimation
Objective: To show the direct change from solid to vapour.
Materials
• Watch glass
• Funnel
• Piece of cotton
• Wet cloth
• Spirit lamp
Procedure and observations
Keep the watch glass on the tripod stand. Put a few crystals of iodine or camphor in the centre of the watch glass and cover it with a funnel (Figure 2). Close the stem of the funnel with a piece of cotton. Wrap a small piece of wet cloth around the stem. Heat the watch glass from below gently with the spirit lamp. Do not use a Bunsen burner. Continue heating till the iodine disappears from the watch glass. Remove the spirit lamp and carefully unwrap the wet cloth and observe the stem of the funnel.
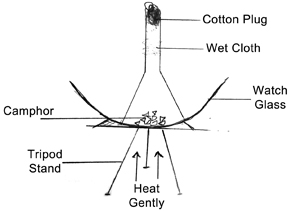
If the experiment is done very carefully with gentle heating, fine crystals of iodine or camphor should be seen deposited on the stem. This depends on the rate of sublimation.
Questions for students
- Why did the iodine or camphor not melt?
- What happens when camphor is burnt?
Note: Iodine would indeed have melted if we raised the temperature above 113° C. However, under the conditions of our experiment we are seeing a direct transition from solid to vapour. (See Reference 1)
C. Chemical changes in matter
In addition to physical changes, chemical changes can also be demonstrated selectively. Such changes can either involve splitting of a compound by supplying energy or displacement reactions, where only electrons are transferred or a chemical reaction forming new compounds releasing heat.
Experiment 4: Decomposition of copper nitrate
Objective: To show substances decompose on heating to give chemically different products.
Materials
• Copper nitrate
• Hard glass test tube
• Bunsen burner
Procedure and observations
Take 2 to 4 g of copper nitrate in the hard glass test tube. Place over the mouth of the test tube a filter paper strip moistened with starch iodide solution. Heat the test tube strongly in the Bunsen flame. Observe the colour of the gas released. Note the colour of the residue. Copper nitrate decomposes to give brown nitrogen dioxide gas which turns the starch iodide paper blue. The residue is black copper oxide.
Note: The colour changes should be explained. The names of the chemical products should also be mentioned. This reaction involves bond breaking and making.
Questions for students
- What was the colour of the gas that was released?
- What was the colour of the residue?
- What happens to the filter paper strip?
Experiment 5: Displacement of copper from copper sulphate solution
Objective: To show a chemical reaction where no bonds are broken or made.
Materials
• Copper sulphate solution
• Zinc pieces
• Glass beaker
Procedure and observations
Weigh about 3g of blue copper sulphate crystal and dissolve it in 100 ml of water in a glass beaker. Stir well to make a homogeneous solution. Next, add a few small pieces of zinc into the same beaker. Let stand for a day. Observe that small particles of reddish copper are formed in the beaker as the zinc dissolves.
Question for students
- What are the reddish particles?
- What happened to the zinc pieces?
Note: This experiment should serve to introduce the electrochemical series in metals to the students. It should also be explained that only electron transfer occurred as copper ions got reduced to metal. Zinc metal got oxidized. No bonds are broken or made.
Experiment 6: Exothermic reaction
Objective: To show that chemical energy is converted to heat energy during a chemical reaction.
Materials
• Quick lime (CaO)
• 250 ml beaker
• Glass rod
Procedure
Take about 10-20 g of quick lime in the beaker. To this add water using a wash bottle slowly stirring the solution during the addition. If possible a thermometer could be used to measure the change in temperature. Note the appearance of the solution.
Question for students
- What is chemical energy?
Note: The teacher could mention that when a chemical reaction takes place, bonds are broken and new bonds are formed. Depending on the overall energy change, heat is either evolved or absorbed. In this case, the bond between calcium and oxygen is broken; the bond between oxygen and hydrogen is broken. Two new bonds are formed.
D. Electrical nature of matter
The electrical nature of matter could be discussed quite early with the students. The well-known ionic nature of salts, the non-ionic nature of substances like water, sugar can be illustrated quite nicely with a few simple experiments. For those students who have already been exposed to the atomic structure, the teacher may perhaps wish to revise and recall the nature of protons, electrons and ions. Given below is an electrical experiment with simple circuitry which the teacher can assemble easily.
Experiment 7: To show conduction of electricity in matter
Objective: To show that some substances conduct electricity while some do not. In addition, this will also demonstrate the importance of the physical state of the substance in order to conduct electricity.
Materials
• Insulated wire
• A small DC or LED bulb rated at 1.5 V
• One AA cell
• One glass beaker
• Distilled water
• Common salt
Procedure and observations
Set up the apparatus as shown in Figure 3. Note that the pieces of wire should be firmly soldered. It is better to use thin flexible wiring used in electronic devices. It is easy to solder them. Also since they are flexible, they will not tip the beaker of water over. Use a higher voltage battery if the bulb does not glow. This experiment must be shown in several steps.
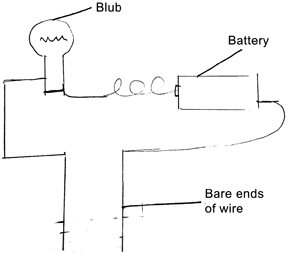
Step 1: First touch the ends of the wires together. The bulb should glow.
Step 2: Dip the ends of the wires into distilled water in the beaker. The amount of water should be sufficient for the wires to dip properly. Now the bulb will not glow, showing that pure water does not conduct electricity. Remove the wires from the water.
Step 3: Add a small amount (about 10 g) of common salt to the water in the beaker. Stir well till all the salt dissolves. Now dip the wires in the solution again. This time the bulb will glow showing that the salt solution is a conductor. (Add more salt if the bulb does not glow).
Step 4: Take a spoonful of common salt in a watch glass. Now touch the ends of the wires after removing from water to the heap of salt. Again the bulb will not glow showing that solid common salt does not conduct electricity.
Questions for students
- In step 1 how did the copper wire conduct electricity?
- Why did the addition of salt to water cause current to flow?
- Why did the current not flow through solid common salt or pure water?
Note: Freely mobile electric charges are required to be present in a substance to make it conducting. Some materials like common salt contain charged particles. These are not mobile in the solid state. Water also does not contain freely mobile charged particles. However, in a salt solution the salt particles are broken down into freely moving ions by the water. Hence, the solution conducts electricity.
Note: Readers who have difficulties in performing any of the experiments above or have queries may contact the author at his email address.
References
- https://doi.org/10.1016/S0187-893X(17)3049-0
- www.middleschoolchemistry.com – Freely downloadable book
The author is a retired teacher of chemistry. He has taught at both the university and school levels. He has some research to his credit. He can be reached at kaone52@gmail.com.
