Vijayalakshmi Nandakumar
This game has been developed to help students learn the chemical symbols for the various elements and to write the correct formula of chemical compounds.
This exercise also helps the young student to learn the valencies for the different elements with ease, without having to learn them by rote. This enhances the student’s skill of observation, quick thinking and recall.
Children of class VII and class VIII can play this game since they will be introduced to the language of chemistry which deals with chemical symbols and formulae.
Materials required
Six or eight cubes of side 2 to 3 cm each. A white paper is pasted on each side of the cube and in bold letters the symbol of the element or radical along with the valency/charge of the respective element or radical is written clearly.
The six sides of three cubes are filled with metallic elements and on the other three cubes, the sides are filled with non-metallic elements or radicals with their charges or valencies.
The symbols should be written clearly such that the child is able to see the letter and the valency/charge on the ion.
Later, as an improvisation, the students can paint that particular side with the colour that is produced by that metallic ion. For example, the side where copper ions is written, can be painted in blue, (as the colour of copper salts are mostly blue) the side where Ferric ion is written can be coloured dark brown to represent rust, ferrous ion to be represented by light green, so on and so forth.
In this manner all the six cubes are prepared and are now ready to use. The cubes can be put in a sac or a box. Now the kit is ready.
Method of playing
The class can be divided into four teams. Then the teacher can ask for a volunteer from each team to take on the challenge in the first round of play. When the players are ready with paper and pencil, the teacher gives the instructions. As soon as the dice/cubes are rolled on to the table, the students are asked to observe the ions/symbols that are on the side facing them. The students are given two minutes to start writing the possible formulae of compounds that can be written using the symbols of the ions seen on the sides of the dice facing them. The teacher keeps an eye on the clock and after two minutes, she asks them to stop writing. Within the two minutes the students will be able to write about a minimum 10 or 15 formulae.
After the students have stopped writing, the teacher checks the list written by each student by reading aloud the formulae written by each student. The common formulae that appear in the list of all four teams are eliminated. The points are calculated for the remaining formulae by adding the valency/charge of each one of the element/ion of each formula of the remaining compounds. This represents the points scored by the respective team.
Example
On the first throw of the cubes, let us say that the symbols facing up on the sides of each cube are
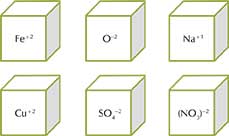 Na+1, Cu+2, Fe+2 [SO4] [NO3] [Oxide]
Na+1, Cu+2, Fe+2 [SO4] [NO3] [Oxide]
The possible formulae that can be seen written by the children are Copper sulphate, Ferrous sulphate, Sodium sulphate, sodium nitrate, sodium oxide, ferrous oxide, copper oxide, copper nitrate and Ferrous nitrate.
If the number of cubes is more, then more permutations and combinations are possible.
The scores are given to each team by adding up the valency/charge of each ion in the compound. For example, if the child has written NaOH, then the Na ion carries 1 point and the OH ion carries 1 point so the total points gained are 2, the points of the formulae not crossed out are calculated and considered as the points scored by the team.
The decision of cancelling the common formulae depends on the teacher. In the initial stages the cancelling need not be done and each group can be allowed to mark their points. Later, when a difficulty level is introduced, the cancellIng of the common formulae can be included for the next level. The aim is not to curb the students’ enthusiasm.
This entire process takes a maximum of six to eight minutes for a single round.
Then the teacher calls for the next set of four volunteers. In this manner the entire class is involved and about four rounds are possible in a 40 minute period.
The teacher can include the other non-players into the game in various ways by inviting a volunteer from each team member to throw the cubes for the respective rounds, to keep track of the points scored by their team, to call out the formulae aloud so that others can check the formulae. The teacher can even ask the students to prepare the symbols to be written on the cube faces prior to the game.
The whole class in that 40 minutes is fully involved in learning the formulae. Once, one set of ions and its valencies are learnt, then another set of ions can be written on those cubes.
The game can be taken one step further to write all the group I metals, group II metals and then group VII and group VI non-metals on the sides of the cubes, and also try to write the possible combination of these metals and non-metals, which will result in the formula of the compounds.
The children themselves can come out with an interesting name for the game. I am sure this exercise will definitely enable them to learn the art of writing correct formulae.
If there are eight cubes then the probability of writing more formulae is increased. The children also come to know the possible combinations that are valid and learn how to name these compounds with the teacher’s help.
Following is a sample list
Metallic elements/ions
Potassium, Lithium, Sodium, Calcium, Magnesium, Ammonium, Zinc, Lead (plumbous), Plumbic, Ferrous, Ferric, Aluminium, Chromium, Hydrogen, Cupric, mercuric, Silver, Cobalt, Nickel.
Non metallic Elements/ions
Oxygen, Flourine, Chlorine, Bromine, Iodine Sulphide Phosphate, Carbonate, Sulphate, Bicarbonate, Sulphite, Nitrate, Nitride, Aluminate, Zincate, Chlorate, hypochlorite, Hydride.
The author is a teacher in Vidyaranya High School, Hyderabad. She can be reached at viji.nandakumar@gmail.com.
