Dr. Jaya Ayyer
As we teach/learn the fundamentals of any subject, it is important to relate the same to everyday life to enable the students to develop a broad based appreciation of the subject. We teach/learn chemistry in the classroom either as a theory of interactions between atoms and molecules, their properties, how they assemble and reassemble through a series of reactions to make new molecules; or as experiments in test tubes, beakers or conical flasks with measurements taken using pipettes, burettes, measuring cylinders etc. At a fundamental level, the understanding one imparts to students through these classroom lessons and lab exercises are very important. However, to appreciate this science and to create a deeper understanding of the role of the same in our lives, one needs to look at its practical applications in a large scale taking into consideration economics, safety, quality assurance and environmental considerations.
Conceptual chemistry taught in the classroom is used by the industry to manufacture the various products we use and relate to in our lives. Removing the misnomers and misconceptions (such as ‘organic’ foods, chemical-free products etc.) regarding ‘chemicals’ and creating clear thoughts in the young minds is also important. This article is aimed at looking at the subject from an industry point of view and is expected to help and encourage the teachers to take the students to a chemical industry to see chemistry in action. Thus, the students can see and understand the importance of studying this subject and also contribute to the same through suitable career options.
How does one define industrial chemistry? How is it different from other traditional branches?
As a chemical scientist who worked initially in academic institutions and then for several years in the chemical industry, I see distinct differences between academic and industrial chemistry, as well as similarities between the two. In the academic approach, one looks at the aspects of chemical transformation, mechanisms of reactions, kinetics and thermodynamics, measurements of various properties and parameters, improvements in reaction yields, elegant synthetic methodologies, understanding of molecular interactions, etc., in a very fundamental manner. This basic understanding further leads to the scale up of practically possible chemistry to large scale transformations keeping in view a variety of factors. Thus, academic and industrial chemistries merge together to mass produce a number of useful products.
If academic chemistry is the basis for industrial chemistry, the foundation for chemical industry is industrial chemistry. The understanding developed through academic chemistry is transformed into a practical science wherein we are able to manufacture the chemicals we need in our daily life. The branches like organic, physical, inorganic chemistries often merge together. The major consideration in industrial chemistry is the economics of production, availability of raw materials, impact on environment, health and safety, and energy conservation. Chemical processes developed in the laboratory are examined from these points of view and scaled up through various steps – bench scale, pilot scale, demonstration scale and finally commercial scale. Depending on individual processes, all these steps may not be strictly required. Increasingly, one makes use of computer aided simulation techniques to scale up processes.
Another interesting aspect is that engineers – chemical, mechanical, instrumentation, etc., have a major role to play. As an industrial chemist, I worked closely with chemical engineers and came to understand and appreciate the scale up and practical considerations and principles involved in taking a process from the laboratory to the plant.
While considering whether a certain chemical process can be scaled up for large scale manufacture of the end product, a number of criteria need to be kept in mind, some of which are:
- Yield of the reactions – if the yields are not high in each step, multi-step processes are unviable.
- Rates of reactions – low rates of reactions lead to very large equipment and high capital cost.
- Use of catalysts – exploring use of catalysts to speed up reactions wherever possible.
- Cost and availability of raw materials – availability of raw materials in large quantities at reasonable cost is very important.
- Energy requirements – high energy cost due to reaction conditions as well as product separation needs can contribute to variable as well as capital cost of production significantly.
- Safety considerations – toxicity of raw materials, products and by-products (solids, liquids, and gases), high temperature and pressure conditions are major safety concerns.
- Material of construction (MOC) – corrosive chemicals (raw materials, catalysts, products, etc.) lead to high cost MOCs leading to high capital cost.
- By product formation and their disposal – quantity and quality of by-products formed, their recycling, reuse and disposal are to be considered.
- Product recovery – can the products be easily separated from the reactants and purified to the required quality in an economic and workable manner?
- Atom efficiency – Major consideration to reduce wastage of costly raw materials and avoid by-product formation.
- Environmental considerations – the need to consider any environmental damages that can be caused and taking corrective action with regular monitoring.
- Cost of production is the most important factor in deciding the successful implementation of any commercial project. While taking into account all the above mentioned points which will have direct or indirect implications on the cost, the viability needs to be worked out.
Using industrial chemistry as foundation, a large number of chemical industries are in existence today. The major ones are:
 Petroleum and petrochemicals industry: This is the most important chemical industry today which converts the fossil based raw materials such as coal, crude oil, and natural gas to basic chemicals and fuels needed for a number of other industries and consumer uses. The petrochemicals are the products derived by breaking carbon – carbon bonds of high molecular weight, hydrocarbons of petroleum and other fossil based fuels.
Petroleum and petrochemicals industry: This is the most important chemical industry today which converts the fossil based raw materials such as coal, crude oil, and natural gas to basic chemicals and fuels needed for a number of other industries and consumer uses. The petrochemicals are the products derived by breaking carbon – carbon bonds of high molecular weight, hydrocarbons of petroleum and other fossil based fuels.
The investments in petrochemical industry run into trillions of dollars. The companies are managed by large multinational corporations who also control a number of downstream chemical companies as well as major polymer industries. Refining of crude oil through cracking and further processing using catalysts is a good example of how the chemical industry has progressed during the last century, both in terms of improvement in better catalytic processes, safety, environmental considerations, energy management, and economies of scale.
The basic chemistry used in the production of petrochemicals is fluid catalytic cracking, hydrogenation followed by high efficiency separation techniques. The important products of this industry are:
- Olefins such as ethylene, propylene and butadiene used as raw materials in polymer, plastics, synthetic fibre, and synthetic rubber industry.
- Aromatics such as benzene, toluene, and xylenes used for production of dyes, detergents, polyurethanes, polystyrenes, and polyterephthalates.
- Synthesis gas, a mixture of carbon monoxide and hydrogen used mainly for production of methanol and ammonia.
- Inorganic chemical industry: Nowadays we think of organic chemicals as the major industrial chemicals, though inorganic chemicals form the oldest chemicals in this respect. Salt, chlorine, caustic soda, soda ash, sulfuric acid, nitric acid, titanium dioxide, and hydrogen peroxide are produced in large tonnage quantities and are essential raw materials for production of many chemicals.
- Synthetic polymer industry: We use a large number of man-made materials in our day-to-day life. In packaging, in consumer products (replacing metals as MOCs), disposable products used in daily life or in medical field, clothing, sports goods, etc. Today it is nearly impossible to imagine our lives without synthetic polymers.
- Fertilizers and agrochemicals: Both these categories of industrial chemicals are important for mass production of food to feed the billions of people of the world. Plants need nitrogen, phosphorous, potassium, sulfur and small levels of micronutrients for their growth. These are supplied through the manufacture of a number of fertilizers using well-established industrial chemistry.
- Ammonia and urea – Ammonia is produced using the Haber-Bosch process from nitrogen and hydrogen using an iron oxide catalyst at high temperature and pressure. Discovered in the early 20th century and the first commercial production achieved in 1913, this continues to be the only process in use till today for large scale ammonia production. Urea is produced from ammonia and carbondioxide.
- Phosphatic fertilizers are produced from mineral rock phosphate (calcium fluorapetite) and a mineral acid such as sulfuric acid or nitric acid. Phosphoric acid produced is suitably neutralized and processed to get granular phosphatic or nitrophosphate fertilizers. Further formulation leads to NPK fertilizers which includes potassium also.
- The term agrochemicals is used to include pesticides, fungicides, herbicides, plant hormones, etc. Complex chemical processes are used for the production of these crop protection chemicals. Due to the inherent toxicity and the hazardous nature of even the raw materials used, the environment damage and residual content of these chemicals in food and water are major concerns.
- Drugs and pharmaceuticals industry: Low volume, high purity and high value chemicals are produced through a variety of industrial chemical processes to get the basic chemicals for the pharmaceutical industry. The relatively smaller scale, conventional, multi-step industrial chemistry used for drug manufacture often produce large amounts of waste. Product purification and separation of optical isomers from racemic mixtures to get the active ingredients also lead to waste or by-products. However, the scenario is now changing as new environmental friendly processes are being developed.
- Dyes, pigments, paints, and coatings: These are high volume chemicals and the chemistry employed for their production has been generating large amounts of hazardous waste which have contaminated rivers and ground water for decades. Due to stringent regulations, improved processes are being developed. Thus, environment friendly processes are the need of the hour for this group of industries for a sustainable future.
- Consumer products: Soaps, detergents, perfumery, cosmetics also form an important part of the chemical industry. While soaps and detergents are high volume, low value chemicals, perfumery and cosmetics are low volume, high value chemicals involving multi-step complex chemical transformations.
The major fertilizers manufactured in large quantities globally are:

For greener, cleaner, atom efficient technologies
The chemical industry is associated with the generation of toxic waste in gaseous, liquid, and solid forms which have been affecting the health and quality of all forms of life on this planet. During the last three decades, the awareness of the long-term impact of the chemical industry has led to stringent pollution control norms. This has led to concerted efforts for developing green or clean technologies on a war footing.
Green industrial chemistry is the minimization or elimination of use, generation and release of chemicals which are hazardous to our environment. The term ‘hazard’ includes threat to safety and health of all living beings in the eco system.
The criterion for evaluation of clean technology for any process is E x Q.
EQ, – Environmental Quotient
E is kg waste/kg product produced
Q is toxicity of the waste or by-product produced.
Many alternate processes have been developed and are already in use. For example, conventional hydrogenation (metal – acid) has been replaced with catalytic hydrogenation, carboxylation using chloride and cyanide followed by hydrolysis has been replaced with direct catalytic cabonylation, etc., in a number of processes. Chiral separation of racemic products has been replaced with enzymatic or chiral catalyst processes wherein only the desired optical isomer is produced.
Atom efficiency concept
The percentage of atom efficiency is defined as:
Molecular weight of desired product x 100
_______________________________________________
Sum total of molecular weights of all reactants
Some examples of commercially used 100% atom efficient processes are:
N2 + 3H2 ——> 2NH3 (catalytic)
2H2 + CO —–> CH3OH (catalytic)
CH3OH+ CO ——–> CH3COOH (catalytic)
CH2=CH2 + H2 ——- > CH3 – CH3 (catalytic)
A visit to a chemical plant
To understand chemistry as practiced on a commercial scale in the industry, a visit to a chemical plant is important. Other than the scale of operation, a major difference one would notice is that the processes are carried out in large vessels and unlike in the laboratory, these are not made of glass (in most cases, though there are a few exceptions). Therefore the chemical processes cannot be directly observed. The progress of any process is monitored by analysis of various parameters. There are continuous process plants and batch process plants.
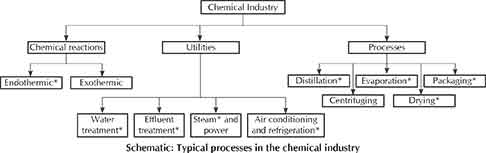
Further, the pipings (often colour-coded) and pumps, storage tanks, utilities like steam, power, cooling systems, etc., separation and purification systems for intermediates, products and by-products, effluent treatment systems before final discharge or disposal of waste and by-products, product packaging and despatch, etc., are integral part of industrial chemistry. The schematic diagram in the previous page shows a typical chemical industry layout.
To truly grasp the import of practising chemistry, it is imperative that one visits a chemical plant.
Summary
Chemistry developed in the laboratory to generate useful products becomes industrial chemistry if the same can be practiced on a commercial scale keeping economics of production, safety, health and environment in mind. The ideal industrial chemistry for sustainable future should meet the following criteria:
1. Atom efficient
2. Safe
3. 100% yield
4. Single step
5. No waste generation
6. Environmentally acceptable
The future of chemical industry will thus be governed by the development of industrial chemical processes which meet the above criteria.
The author is Additional General Manager (Retd.), Gujarat Narmada Valley Fertilizers & Chemicals Ltd. Bharuch, Gujarat. She can be reached at jayayyer@yahoo.com.